Leucine has long been recognized as one of the most metabolically important essential amino acids, particularly for its role in stimulating muscle protein synthesis by activating the mTOR pathway. However, emerging research from the University of Cologne highlights a second, fundamentally different role for leucine—one that operates directly at the level of mitochondrial function and cellular energy production.
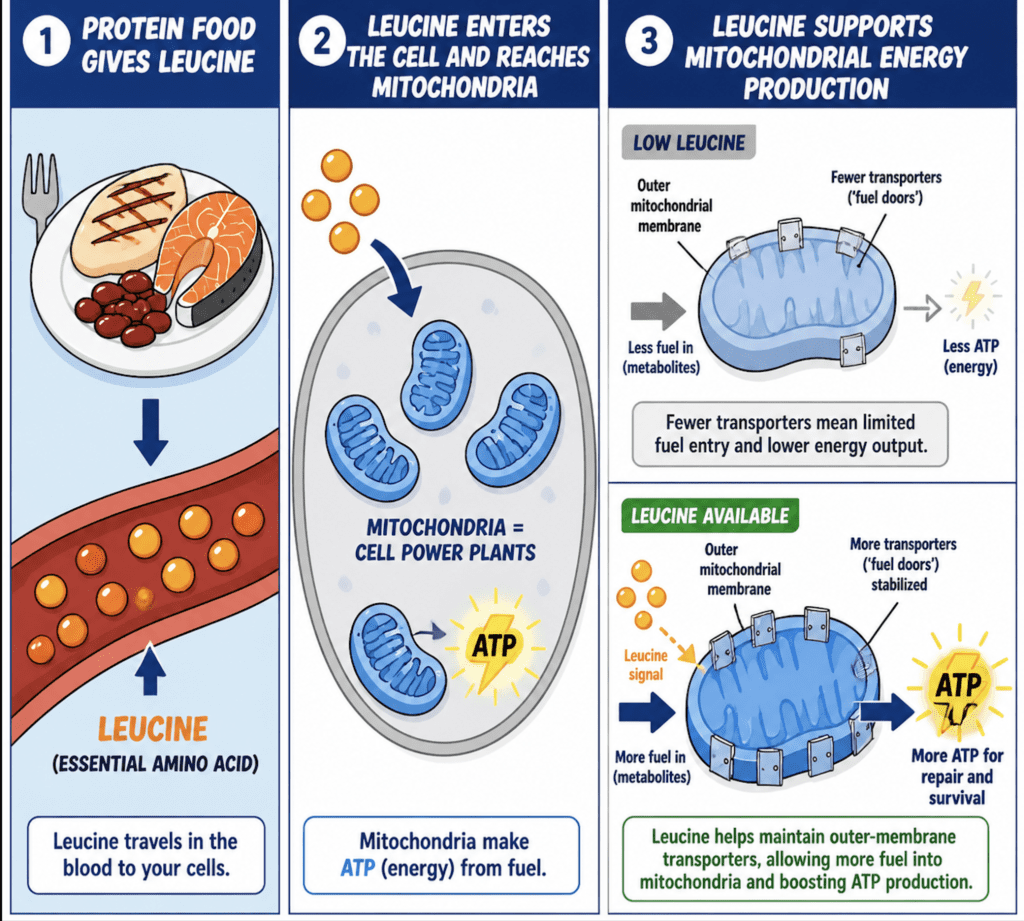
Mitochondria generate ATP, the cell’s primary energy currency, through oxidative phosphorylation. This process depends not only on internal mitochondrial enzymes but also on a carefully regulated exchange of metabolites across the outer mitochondrial membrane. This membrane contains specialized transport proteins that act as controlled gateways, allowing substrates such as pyruvate, fatty acids, and other intermediates to enter the mitochondria, while exporting metabolic byproducts.
The new findings show that leucine plays a key role in stabilizing these outer mitochondrial membrane proteins. Under normal physiological conditions, cells maintain protein quality through a surveillance system called endoplasmic reticulum–associated degradation (ERAD). Within this system, a protein complex involving SEL1L identifies misfolded or damaged proteins and targets them for degradation. While this process is essential for preventing the accumulation of dysfunctional proteins, it can also remove proteins that remain functionally important, particularly under conditions of metabolic stress or high demand.
Leucine appears to modulate this quality-control pathway. Specifically, it suppresses SEL1L activity, thereby slowing the degradation of certain mitochondrial membrane proteins. By preserving these transport proteins, leucine enhances the efficiency of metabolite flux into the mitochondria. The net effect is increased mitochondrial respiration and ATP production, particularly during periods of nutrient availability when cells are primed for growth, repair, or increased functional output.
This mechanism adds a new dimension to leucine’s role as a nutrient signal. In addition to activating mTOR and promoting anabolic processes, leucine now appears to coordinate energy supply by directly supporting mitochondrial infrastructure. In essence, it helps ensure that when a cell receives signals to grow or repair, the necessary machinery for energy production is simultaneously reinforced.
The pathway also involves GCN2, a nutrient-sensing kinase that typically responds to amino acid deprivation. Under low-amino-acid conditions, GCN2 is activated and helps initiate adaptive responses that conserve resources. Leucine availability suppresses this stress response, further promoting a metabolic state geared toward energy production and biosynthesis. The interplay among leucine, GCN2, and SEL1L creates a regulatory axis linking nutrient sensing to mitochondrial performance.
Experimental validation of this mechanism comes from both organismal and cellular models. In Caenorhabditis elegans, disruption of leucine metabolism led to impaired mitochondrial respiration and a measurable decline in fertility, underscoring the importance of efficient energy production for reproductive and systemic health. In human lung cancer cell lines, alterations in leucine metabolism were associated with enhanced survival, particularly under conditions of metabolic stress. This suggests that cancer cells
These findings have significant implications for several areas of medicine. In regenerative medicine, mitochondrial function is a central determinant of stem cell viability, differentiation capacity, and engraftment success. Cells introduced into damaged or ischemic tissues must rapidly adapt to hostile environments with high energy demands and variable nutrient availability. Enhancing mitochondrial efficiency through pathways such as the leucine–GCN2–SEL1L axis could theoretically improve cell survival and therapeutic outcomes.
Similarly, in conditions characterized by mitochondrial dysfunction—such as metabolic syndromes, neurodegenerative diseases, and age-related decline targeting this pathway could offer a novel strategy to restore energy balance. By stabilizing mitochondrial membrane proteins and improving substrate utilization, it may be possible to enhance cellular resilience without directly manipulating mitochondrial DNA or enzyme systems.
At the same time, this mechanism introduces important safety considerations. The suppression of protein degradation pathways, even selectively, raises the possibility of impaired proteostasis over time. Accumulation of damaged or improperly folded proteins can contribute to cellular dysfunction, particularly in long-lived cells such as neurons. Moreover, the observation that cancer cells may benefit from enhanced leucine signaling highlights the need for caution, especially in patients with active or high-risk malignancies.
Another key limitation is that current evidence is derived from preclinical models, including nematodes and cultured human cells. While these systems provide strong mechanistic insight, they do not fully replicate the complexity of human physiology. Factors such as tissue-specific metabolism, systemic hormone signaling, and long-term regulation of nutrient pathways remain to be clarified in clinical settings.
In a broader sense, this research reframes how we think about nutrients. Rather than serving solely as substrates or fuel, molecules like leucine act as regulators of cellular systems, capable of fine-tuning energy production, stress responses, and structural integrity. This positions amino acids not just as building blocks, but as dynamic participants in cellular decision-making.
Ultimately, the identification of leucine’s role in stabilizing mitochondrial membrane proteins provides a new conceptual link between nutrition, metabolism, and cellular performance. It opens the door to more targeted approaches in regenerative medicine and metabolic therapy, while also emphasizing the importance of context, dosing, and safety in any future clinical application.
Dr. P