Here is a diagram of a free radical:
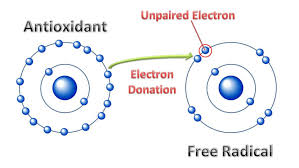
We can see that the free radical has an unpaired electron. Oxidative stress occurs when an oxygen molecule splits into single atoms with unpaired electrons, which are called free radicals. The body is under constant attack from oxidative stress. Oxygen in the body splits into single atoms with unpaired electrons. The “unpaired” electrons become unstable free radicals that seek other atoms or molecules to bond to. If this continues to happen, it begins a process called oxidative stress. How the body deals with this oxidative stress can determine if one is healthy or one is prone to a variety of degenerative medical conditions.
FREE RADICALS: SOMETIMES AN ENEMY AND SOMETIMES A FRIEND OF THE BODY
Free radicals damage the growth, development and survival of cells in the body. Their reactive nature allows them to engage in unnecessary side reactions causing cellular impairment and eventually injury when they are present in disproportionate amounts. They directly impair cell membranes and DNA. The next diagram shows various other causes of free radicals but utilization of oxygen is the most common cause.
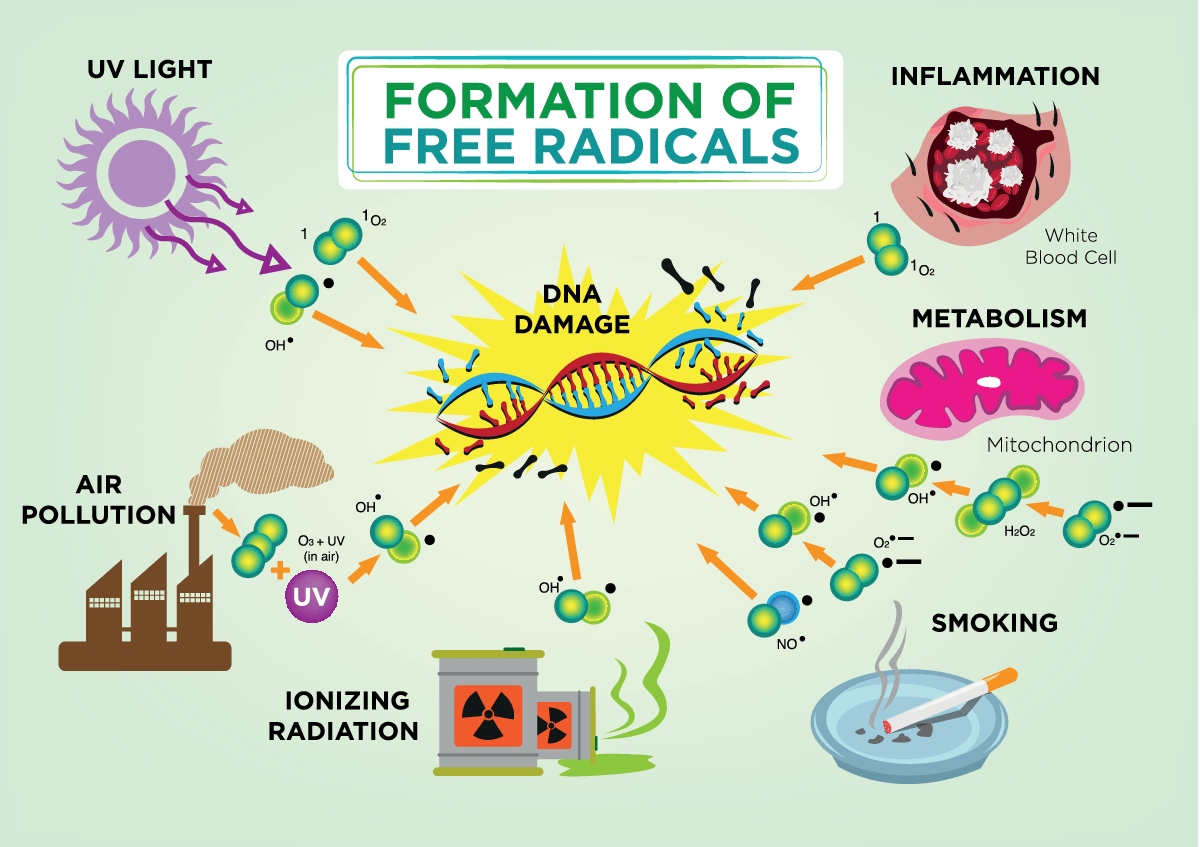
We can see that Free radicals and other ROS (Reactive Oxygen Species) are derived either from normal essential metabolic processes in the human body or from external sources such as exposure to X-rays, cigarette smoking, air pollutants, and industrial chemicals. The bottom line is that even if you live in a pristine area with no pollution your body will still be bombarded by free radicals since you must BREATHE OXYGEN which produces free radicals. Disease processes can also result in overproduction of oxidants. Infections, allergies, and autoimmune diseases activate our immune cells, which produce reactive oxidants in order to kill germs that the immune system assumes to be present, but our otherwise healthy cells get caught in the cross-fire and sustain collateral damage that we see and feel as inflammation. Other major diseases associated with aging, such as heart attacks, stroke, cancer, and neurodegenerative conditions such as Alzheimer’s disease also increase production of oxidants, creating oxidative stress, and inflammation. One further free radical body that needs mentioning is a Reactive Nitrogen Species. Actually, both Reactive Oxygen Species (ROS) and Reactive Nitrogen Species (RNS) can have duel effects especially the RNS. Recent evidence has suggested that intracellular RNS are an important component of intracellular signaling cascades. ROS can have similar effects in that they can affect a wide variety of metabolic functions in the body. RNS can also be very beneficial in fighting certain bacterial and viral infections in the body.
Further elevation of RNS leads to fatigue. RNS can induce mitochondrial biogenesis, influence the levels of SIRT1 which controls vital metabolic processes, and is involved in hypertrophy of skeletal muscle. Massive elevation of RNS can lead to increased oxidative stress and alteration of lipids, proteins, and DNA. It is suggested that a moderate level of oxidative damage of lipids can be important to membrane remodeling. A moderate level of protein modification by RNS could be an important control of protein synthesis. Moreover, moderate level of oxidative DNA modification could be important to increased gene expression and cellular signaling. In skeletal muscle the age-associated decline in muscle mass and function is also related to RNS level and inflammation. Regular exercise has a powerful effect on RNS production, antioxidant, and oxidative damage repair systems. Moreover, regular exercise can attenuate the age-associated function deteriorations.
ANTIOXIDANTS
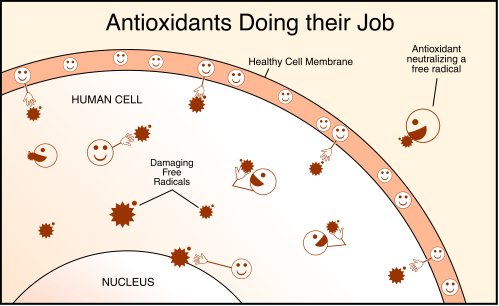
Before we move on, when discussing oxidative stress, we must also mention ANTIOXIDANTS. Antioxidants are substances that can prevent or slow damage to cells caused by free radicals. They are sometimes called “free-radical scavengers”. Unfortunately, the human body is not so cut and dry when it comes to dealing with free radical generation. Oxidants are produced by the process of “burning” the foods that sustain us, just as the internal combustion engines in our cars produce similar reactive chemicals. Cars have catalytic converters to neutralize and detoxify these reactive chemicals; our bodies have antioxidant enzymes to accomplish the same result. Realize that we can supply our bodies with anti-oxidants from a dietary source and at the same time the body is able to produce powerful anti-oxidant compounds on its own. The following diagram shows anti-oxidants at work.
WHAT IS THE TRUE SCIENCE OF OXIDATIVE STRESS?
Remember that we said ROS and RNS can act as signaling molecules in the body. This is where the true science comes into the play. As was mentioned, these two free radicals can act as signaling molecules. Although it may seem hard to fathom, some oxidative stress is actually a “good” thing. There are two main pathways in the body that control inflammation. These two pathways are called the NrF2 (nuclear factor erythroid 2–related factor 2) pathway and the NFkB (Nuclear Factor kappa-light-chain-enhancer of activated B cells) pathway. These pathways are at the extremes of the spectrum. These pathways are actually transcription factors. Transcription factors are proteins that help turn specific genes “on” or “off” by binding to nearby DNA. Transcription factors that are activators boost a gene’s transcription such as the case with Nrf2. While repressors decrease transcription as is the case with NFkB.
The amount of oxidative stress will determine which pathway will be activated. This can mean the difference between health and disease. Think of Nrf2 as a thermostat of inflammation. Nrf2 has traditionally been regarded as the cell’s main defense mechanism and a major regulator of cell survival. Activation of the Nrf2 defense response has been shown to protect against neuro-degenerative diseases, aging, diabetes, photo-oxidative stress, cardiovascular disease, inflammation, pulmonary fibrosis, acute pulmonary injury, and cancer. Mild stress will activate this pathway. Nrf2 will initially be found in the cytoplasm of the cell. When in the cytoplasm it is tightly bound to Keap 1. Under quiescent conditions, Nrf2is anchored in the cytoplasm to Keap1. Keap 1 is a protein which binds the Nrf2 which is also a protein. Keap 1 keeps the Nrf2 molecule under wraps until a stimulus is received (for instance high levels of free radicals) and Nrf2 breaks its chains and goes into the cell nucleus where it stimulates certain genes to produce biochemical compounds. These compounds are called ARE which stands for Antioxidant Response Elements. These are very potent anti-oxidants that the body produces. There are a number of them.
The following diagram shows the Nrf2 pathway at work:
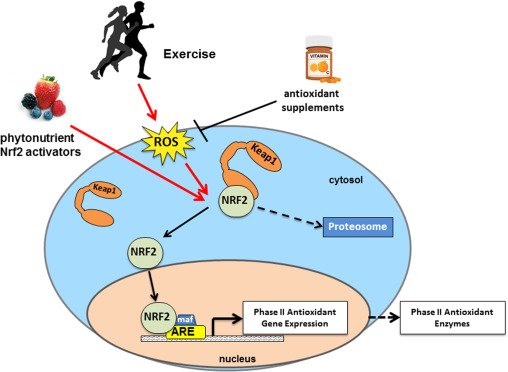
We are able to see the Nrf2 is held in the cytoplasm by the KEAP1 protein. Subsequently, with activation the Nrf2 makes its way into the nucleus and subsequently affecting the nuclear DNA thus acting as a transcription factor turning on certain genes and subsequently producing the ARE products. The Nrf2 pathway senses the need for these antioxidant enzymes and regulates their production to maintain metabolic balance. Several things can upset this delicate balance, and simply growing older is one of them. We produce less Nrf2 as we age, and the balance slowly tips toward the oxidative side—a condition known as “oxidative stress.” Disease processes can also result in overproduction of oxidants. Infections, allergies, and autoimmune diseases activate our immune cells, which produce reactive oxidants (O2-., H2O2, OH. and HOCl) in order to kill germs that the immune system assumes to be present, but our otherwise healthy cells get caught in the cross-fire and sustain collateral damage that we see and feel as inflammation. Other major diseases associated with aging, such as heart attacks, stroke, cancer, and neurodegenerative conditions such as Alzheimer’s disease also increase production of oxidants, creating oxidative stress, and inflammation. The bottom line for the Nrf2 pathway is that some oxidative stress is good for activating this pathway, while too much will turn it off.
Some final thoughts on the Nrf2 pathway. Nrf2 has a crucial role in maintaining cellular redox homeostasis. Nrf2 affects the mitochondrial membrane potential and ATP synthesis. Nrf2 influences mitochondrial fatty acid oxidation. Nrf2 supports the structural and functional integrity of the mitochondria. Nrf2 activators have beneficial effects when mitochondrial function is compromised. Anything that increases mitochondrial health will increase our health.
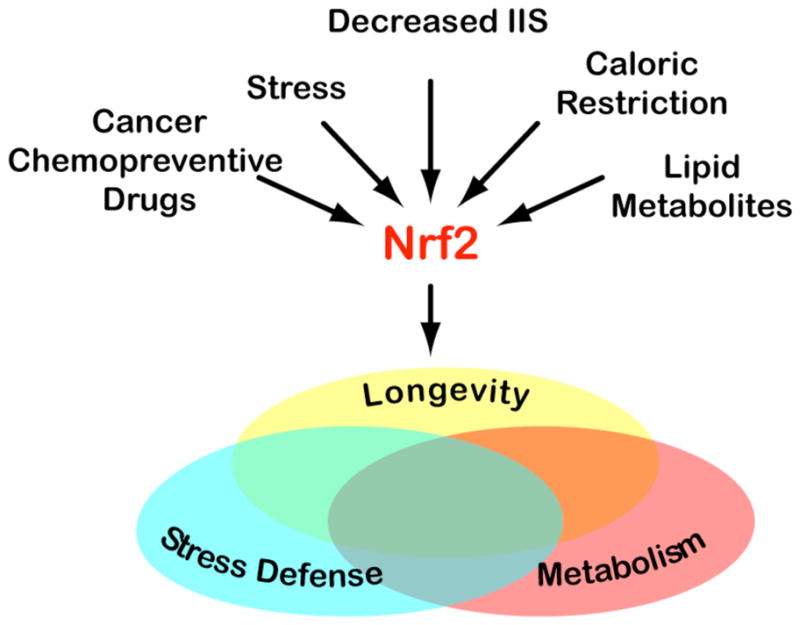
The next diagram shows some other methods of stimulating this pathway:
OZONE THERAPY
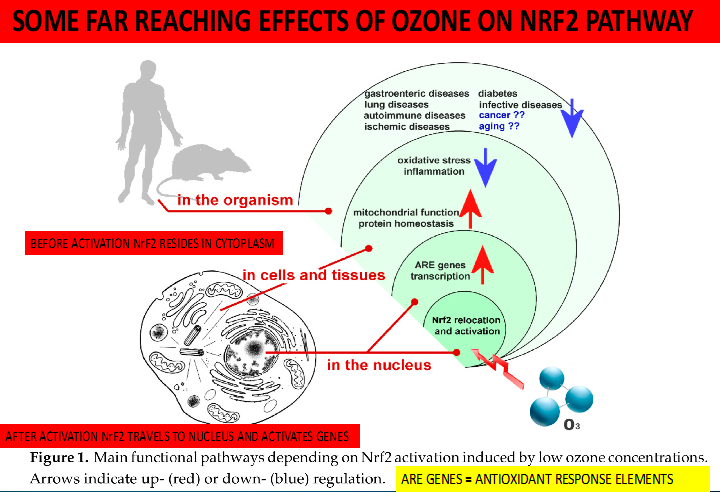
One very effective method of stimulating the Nrf2 pathway is thru the use of Medical Ozone gas. The method is called EBO2 which stands for Extra Corporeal Blood and Ozone therapy. This is a cutting-edge technique where blood is both filtered and subsequently Ozonated after removing a number of offending agents. The Medical Ozone gas when delivered in this manner is a potent stimulator of the Nrf2 pathway as can be seen I the following illustration:
THE NFkB PATHWAY: THE ANTITHESIS OF THE Nrf2 PATHWAY
Whenever we discuss oxidative stress we must also address the “elephant in the room” namely the NFkB pathway. NF-kB is a short name of Nuclear Factor kappa-light-chain-enhancer of activated B cells. It is not a single protein, but a small family of inducible transcription factors that play an important role in almost all mammalian cells. NF-kB acts as a central mediator of immune and inflammatory responses, and is involved in stress responses and regulation of cell proliferation and apoptosis. The respective NF-kB target genes allow the organism to respond effectively to these environmental changes. The classical NF-kB pathway is activated by a variety of inflammatory signals, resulting in coordinate expression of multiple inflammatory and innate immune genes. Nuclear factor-κB is essential for inflammatory responses. It is one of the most important molecules linking chronic inflammation to cancer while its activity is tightly regulated by several mechanisms. As we can see from the following diagrams there are a number of methods in which the NFkB pathway gets stimulated:
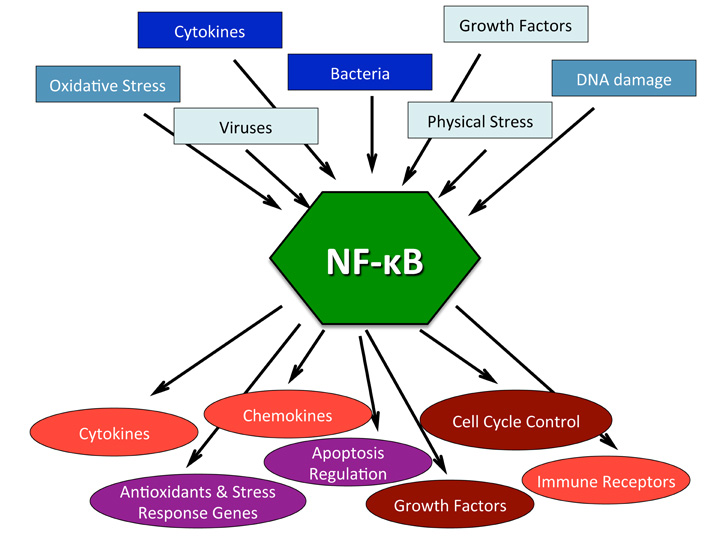
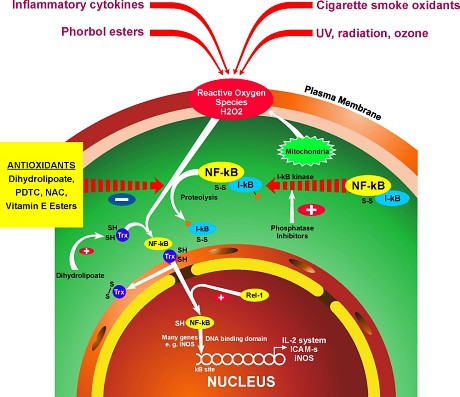
We can see from the two proceeding diagrams that NFkB pathway is stimulated either by external or internal factors. NF-κB is a master switch that sets in motion a chain of events that is specific to a cell type and its environment. Some auto-immune diseases (e.g., arthritis, type 1 diabetes, asthma, Crohn’s disease and irritable bowel) are associated with an excess of NF-κB. Its activation generally rises with age, but it is necessary at all ages, particularly for its contribution to the regulation of apoptosis (the selective elimination of cells that are potentially damaging). It will probably be necessary to strongly but selectively inhibit NF-κB, beginning in middle age.
Inflammatory responses are complex and focused on the immediate threat at hand. We must remember that NFkB is not always a culprit. It can actually save our lives.
The transcription factor NF-κB regulates multiple aspects of innate and adaptive immune functions and serves as a pivotal mediator of inflammatory responses. NF-κB induces the expression of various pro-inflammatory genes, including those encoding cytokines and chemokines. It also participates in inflammasome regulation. An inflammasome is a multiprotein intracellular complex that detects pathogenic microorganisms and sterile stressors. It subsequently activates the highly pro-inflammatory cytokines interleukin-1 and interleukin 18.
In addition, NF-κB plays a critical role in regulating the survival, activation and differentiation of innate immune cells and inflammatory T cells. This can be instrumental in dealing with a bacteria or virus. However, deregulated NF-κB activation contributes to the pathogenic processes of various inflammatory and autoimmune diseases. It can also be responsible for some of the complications we are seeing with Covid-19 such as a cytokine storm. In a cytokine storm in the body’s attempt to eliminate the pathogen there is much collateral damage to surround tissue.
WHAT ARE THE BEST METHODS THAT WE CAN USE TO STIMULATE THESE PATHWAYS IN ORDER TO ASSIST THE BODY WITH OXIDATIVE STRESS.
The real key is to activate the Nrf2 pathway and quite down the NFkB pathway. Nrf2 contributes to the anti-inflammatory process by orchestrating the recruitment of inflammatory cells and regulating gene expression through the antioxidant response elements (ARE). It regulates anti-inflammatory gene expression and inhibits the progression of inflammation. There is a regulatory loop of Nrf2 and NFĸB. In other words when Nrf2 is stimulated then stimulation of NFkB diminishes.
OF ALL METHODS TO INCREASE THE STIMULATION OF THE NRF2 PATHWAY, WE FEEL THAT MEDICAL OZONE DELIVERED VIA THE EBO2 PROTOCOL TO BE ONE OF THE MOST EFFECTIVE METHODS.
In addition to the EBO2 technique, a number of supplements seem to be noteworthy. These include trans-pterostilbene, TrueBroc broccoli extract, Turmeric extract (curcumin) “Ultra-cur”, Green Tea Extract, Resveratrol, Fumaric acid compounds= potent stimulator of nrf2, Quercetin, Genistein (soy isoflavones), NAD, Neo-40 a nitric oxide stimulator. A Fumaric acid derivative was just recently approved by the FDA for the treatment of certain forms of Multiple Sclerosis.
One final note, there is a company called Clearwater Nutrition Lab which will soon launch a product to address the Nrf2 pathway. This product has many of the above compounds. The reason I know this is that I helped to design it for them.
-JP